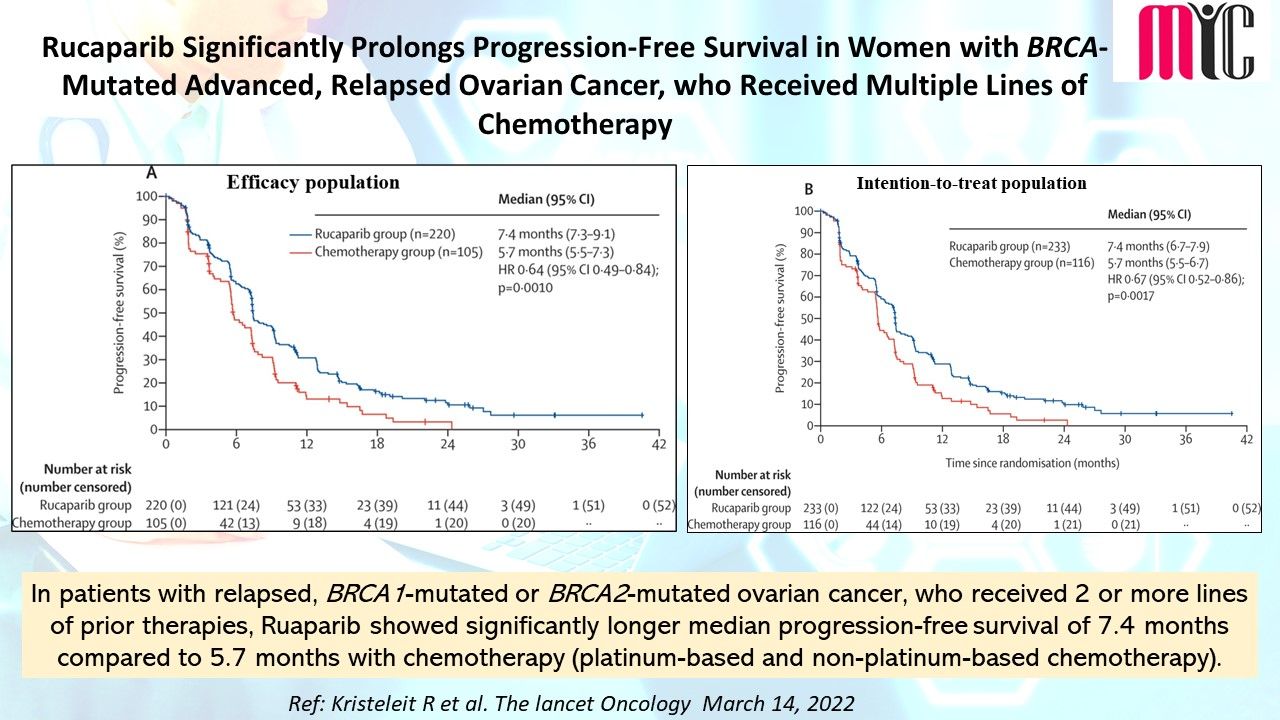
26 Mar Rucaparib significantly prolongs PFS compared with chemotherapy in women with BRCA-mutated advanced, relapsed ovarian cancer, who received 3 or more lines of prior therapies
In patients with relapsed, BRCA1-mutated or BRCA2-mutated ovarian cancer, who received 2 or more lines of prior therapies, Rucaparib showed significantly longer median progression-free survival of 7.4 months compared to 5.7 months with chemotherapy (platinum-based and non-platinum-based chemotherapy). # ARIEL4 study. (Ref: Kristeleit R et al. The lancet Oncology March 14, 2022)
#oncologyresearch
https://www.linkedin.com/feed/update/urn:li:activity:6913402413664104448